
Unravelling the impact of ICH E6(R3) on Good Clinical Practice
Good Clinical Practice (GCP), the bedrock of ethical and high-quality …
Newsletters and Deep Dive digital magazine

Good Clinical Practice (GCP), the bedrock of ethical and high-quality …
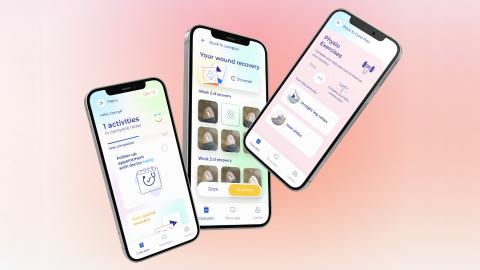
Hospital staff in the UK have developed and piloted an app that can …

A personalised vaccine for the aggressive and fatal brain cancer …

Novo Nordisk’s breakneck sales growth has continued in the first …

A business is only as successful as its people - and the life …

Accelerate your capsid and payload design with the 4th Next …

Astellas has clearly been impressed with a partnership signed last …

WASHINGTON, D.C., April 30, 2024 –The Food and Drug Administration’s …

Jacobio Pharma has reported the results of its pivotal Chinese study …

The UK healthcare system is confronting unprecedented challenges due …

GSK looks like it is starting to show the benefit of a revamp of its …
Polish drug delivery company Biotts says it has demonstrated delivery …

Pfizer will draw on the expertise of Citi analyst Andrew Baum when it comes to portfolio management and capital deployment, naming him chief strategy and innovation office


Explore “Innovation as Strategy” framework, providing a guide for employing innovative strategies in life sciences.

In this edition of Deep Dive, Unpacking complex choreography in the biologics patent dance, Alexion examines the urgent need for accelerated access programmes for rare disease treatments, and Putna

It will come as no surprise to anyone in the healthcare space that doctors today are under an inordinate amount of pressure.

We are living in an era of unprecedented advances in cancer treatments with transformative therapies being regularly approved.

Accelerate your capsid and payload design with the 4th Next Generation Gene Therapy Vectors Summit (June 12-14, Boston), the industry’s only hub for covering the latest advances and innovations in

WASHINGTON, D.C., April 30, 2024 –The Food and Drug Administration’s (FDA) final rule has reclassified laboratory developed tests (LDTs) as medical devices, according to Avalere.